|
Sugar in the form of blood glucose provides
essential energy for cells. When its usual dietary
source — carbohydrates — is scarce, the liver can
produce it with the aid of fat, but new research
from Johns Hopkins now adds to evidence that other
tissues can step in to make glucose when the liver’s
ability is impaired, and that the breakdown of fats
in the liver is essential to protect it from a
lethal onslaught of fat.
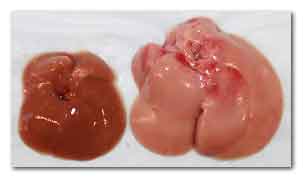
The livers of a normal mouse (left) and a mouse
whose liver cells lack Cpt2 (right) after eating a
high fat diet.
Image credit: Courtesy of Cell Press
“We were surprised that other tissues, including the
kidney and intestine, could compensate so well when
the liver’s ability to generate glucose is
impaired,” says Michael Wolfgang, Ph.D., “but, then
again, it’s not unusual in biology to have backup
systems for something so crucial to survival as
providing energy to cells.”
Wolfgang, an associate professor of biological
chemistry at the Johns Hopkins University School of
Medicine, says that according to textbooks, the
ability to maintain blood glucose during starvation,
known as gluconeogenesis, requires the breakdown and
processing of fatty acids, known as fatty acid
oxidation.
Also, it is thought that about 90 percent of
gluconeogenesis occurs in the liver, while the other
10 percent occurs in the kidneys and gut. So when he
and his team deleted the gene Cpt2, which is
necessary for fatty acid oxidation, from mouse liver
cells, they didn’t expect them to survive without a
continual supply of carbohydrates.
“Mice live for the first two weeks on milk from
their moms, which is high in fat and low in carbs,”
says Wolfgang, “so we were surprised that they did
OK when their liver’s ability to burn fat to make
glucose and ketones was crippled.”
Wolfgang explains that when enzymes break down
high-energy compounds — fatty acids — they produce
multiple molecules of acetyl CoA, which funnel into
two different reactions.
One generates energy-containing molecules of ATP,
which can be used to make glucose to maintain blood
glucose levels in animals that haven’t eaten
carbohydrates in a while.
The other reaction makes molecules called ketones,
which can be used by some tissues, like the brain,
as an alternative energy source when glucose is
scarce.
The researchers were also surprised that the mice
lacking Cpt2 in the liver weighed the same as normal
mice and used the same amount of energy. They even
used comparable amounts of fat and sugar as fuel
sources. The only apparent change was lower levels
of circulating ketones, which was expected.
“I still find it hard to wrap my head around how
these seriously compromised mice not only survived
but were indistinguishable from normal mice in their
energy use,” says Wolfgang.
In further tests, when the researchers examined the
mouse kidneys, they found an increased fat content
and the genes responsible for fatty acid oxidation
were more active, suggesting that the kidney had
dialed up the process compared to normal mice.
Those results begged the question of just what
distress signals the liver was sending to tell other
tissues to help.
Examining the activity of genes in the liver, the
team found huge changes, including in some
long-range signaling molecules.
One, known as FGF21, caught their eye because it
encourages cells to absorb carbohydrates and break
down fats, and is being tested as a treatment for
diabetes and obesity. Indeed, they found its levels
greatly elevated in the blood of mice whose livers
lacked the ability to burn fat.
To find out how fasting would affect fatty acid
oxidation in the liver, the researchers withheld
food from the genetically modified mice for 24
hours. But the mice were able to adjust to even this
challenge. Their overall energy usage was normal, as
were their blood glucose levels, though their livers
were fatty and they had too many circulating fats
and no circulating ketones. The researchers also saw
changes in gene activity levels related to
oxidation, both in the liver and the kidney.
To better understand the unique metabolism of the
mice lacking Cpt2, the researchers next put them on
a high-fat, “ketogenic diet,” similar to the
commercial Atkins diet that is very low in
carbohydrates.
Although, according to Wolfgang, they were consuming
a lot of calories and essentially eating butter for
every meal, their livers couldn’t handle the fat,
and the diet was eventually lethal to the mice.
The mice had seemingly dissolved all fat tissue
throughout their bodies, but their livers were
engorged with fat molecules. Wolfgang explains that
fat tissue throughout the body breaks down fats into
fatty acids, which are then sent to the liver for
processing.
Wolfgang says: “The liver knew it needed to burn fat
to make glucose, so it kept asking fat tissue to
send fatty acids. But it couldn’t burn those fatty
acids, so it just absorbed them and got too fat to
function.”
Wolfgang says the team’s data suggest that almost
all circulating ketones are produced by the liver
through fatty acid oxidation. Ketones are known to
slow the breakdown of fats in fat tissue, so their
absence in the mice probably contributed to the
continued onslaught of fats on the liver.
All of this, says Wolfgang, might help explain how
and why metabolism goes haywire in people who are
obese, diabetic or are born with genetic errors that
affect fatty acid oxidation, including errors in
Cpt2, which can be lethal.
Wolfgang also points out that what acutely threatens
people with type 1 diabetes is a condition called
ketoacidosis. Since these individuals lack insulin,
which cells need to absorb carbohydrates, their
cells end up relying too heavily on fatty acid
oxidation in the liver, which generates ketones. Too
many ketones in the blood make it acidic, which
decreases its capacity to carry oxygen. Wolfgang
hopes that further studies to understand how the
body adjusts to a compromised liver will shed light
on how to prevent ketoacidosis and better regulate
or re-regulate faulty metabolism.
Other authors of the report include Jieun Lee,
Joseph Choi and Susanna Scafidi of the Johns Hopkins
University School of Medicine.
For more information
Cell Reports
Hepatic Fatty Acid Oxidation Restrains Systemic
Catabolism during Starvation
Link...
Johns Hopkins University
Link...
MDN |