|
Gut microbes “say” influences the activity of a key
clock-driven protein called NFIL3, which can set
intestinal cells up to absorb and store more fat
from the diet while operating at hours that might
run counter to our fixed biological clocks.

NFIL3 is a transcription factor, a protein that
switches certain genes on and off.
Earlier studies had focused on its role in immune
cells, but a team led by Lora Hooper at the
University of Texas Southwestern Medical Center,
Dallas, discovered that NFIL3 is also found in cells
in the inner lining, or epithelium, of the mouse
small intestine.
Intriguingly, as reported recently in the journal
Science, they noticed that NFIL3 levels were much
lower in the intestines of “germ-free” mice that
don’t have any gut microbes.
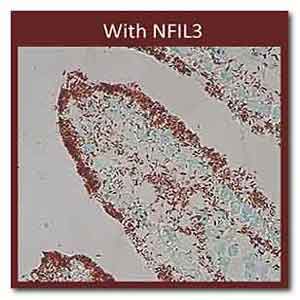
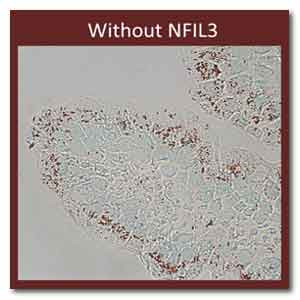
IMAGE Caption: Lipids (red) inside mouse intestinal
cells with and without NFIL3. Credit: Lora V.
Hooper, University of Texas Southwestern Medical
Center, Dallas
Hooper and colleagues generated mice that lacked the
Nfil3 gene only in epithelial cells.
When those animals were raised on normal mouse chow,
they grew leaner than their normal littermates.
When both groups were placed on a high-fat diet, the
mice lacking NFIL3 in their intestines packed on a
lot less weight.
They also had lower body fat and other indications
of better health, including lower blood lipids, less
fat in the liver, and fewer early signs of diabetes.
Many metabolic pathways are synchronized to
day-and-night cycles that oscillate between greater
activity during daylight and reduced activity at
night.
Orchestrating this 24-hour cycle is a core network
of transcription factors called the circadian clock
that modulate the expression of numerous genes,
including Nfil3, over the course of the day.
The circadian clock, however, doesn’t operate in
isolation.
Recent studies suggest gut microbes interact with
these transcription factors to affect metabolism
profoundly.
The researchers established that the absence of gut
microbes in germ-free mice caused NFIL3 levels to
flatten out, losing the protein’s normal 24-hour
rhythm and suggesting its expression is dependent on
the microbes.
What might this mean for fat storage and weight
gain?
For clues, the researchers delved deeper into the
basic biology, comparing the activity of other genes
in intestinal cells with and without NFIL3.
They uncovered differences in the activity of 33
genes, many of which follow a regular daily rhythm.
Seventeen of those genes were already known to
encode proteins involved in the uptake of lipids or
other aspects of metabolism.
It looked as though mice lacking NFIL3 might be
leaner because their intestinal cells take up and
store less fat from the diet.
To nail down this point, the researchers used a red
stain to visualize lipids in the animals’
intestines.
As shown above, intestinal cells lacking NFIL3
contained much less lipid and the extra fat passed
right on through the digestive systems of the mice.
Further studies showed gut microbes don’t talk
directly to intestinal cells and NFIL3.
Rather, they go through an intermediary, sending
messages to intestinal cells via the immune system.
Of course, these findings are in mice. But humans
are known to have NFIL3, too, and further research
will be needed to chase down this potentially
important lead.
NFIL3 is a pleiotropic transcriptional regulator in
both acquired and innate immunity. It has a critical
role in T helper-2 (Th2) cytokine gene expression
and controls IgE class switching and natural killer
(NK) cell developmentNFIL3 is a pleiotropic
transcriptional regulator in both acquired and
innate immunity. It has a critical role in T
helper-2 (Th2) cytokine gene expression and controls
IgE class switching and natural killer (NK) cell
development
Link...
OMIM® - Online Mendelian Inheritance in Man®
is a comprehensive, authoritative compendium of
human genes and genetic phenotypes that is freely
available and updated daily.
The full-text, referenced overviews in OMIM contain
information on all known mendelian disorders and
over 15,000 genes.
OMIM focuses on the relationship between phenotype
and genotype. It is updated daily, and the entries
contain copious links to other genetics resources
Link....
For more information
U.S. National Institutes of Health
Link...
The intestinal microbiota regulates body composition
through NFIL3 and the circadian clock.
Link...
Antagonistic role of E4BP4 and PAR proteins in the
circadian oscillatory mechanism.
Link...
Transkingdom control of microbiota diurnal
oscillations promotes metabolic homeostasis.
Link...
MDN |