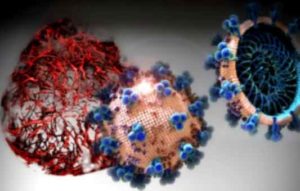
In cell cultures analyzed in the current study, hrsACE2 inhibited the coronavirus load by a factor of 1,000-5,000. Credit: IMBA/Tibor Kulcsar
A drug already tested against lung disease could potentially inhibit COVID-19 by reducing the coronavirus load that enters the lungs and other organs.
The drug, called APN01, is developed by Aperion Biologics and has just received regulatory approvals for the treatment of 200 COVID-19 patients in Austria, Germany and Denmark in a phase-II study.
That is according to a study in human cell cultures and organoids by researchers at Karolinska Institutet in Sweden and the University of British Columbia (UBC) in Canada, published in the journal Cell.
In the study the researchers used tissue samples from a patient with COVID-19 to isolate and cultivate SARS-CoV-2, the virus that causes the disease COVID-19.
In cell cultures, they were able to show how the spike protein in SARS-CoV-2 binds to a cell surface receptor called angiotensin converting enzyme 2 (ACE2) in order to enter our cells.
It is the same mechanism that the original SARS-virus from 2003 used to bind to our cells, and which has been described by several of the researchers in previous studies.
By adding a genetically modified variant of this protein, called human recombinant soluble angiotensin-converting enzyme 2 (hrsACE2), the researchers wanted to test if the virus could be stopped from infecting the cells.
The result now published shows that hrsACE2 reduced viral growth of SARS-CoV-2 by a factor of 1,000 to 5,000 in cell cultures.
The result was dose dependent, meaning it varied depending on the total amount of virus in relation to the total amount of hrsACE2.
The authors were also able to verify these data from regular cell cultures in engineered miniature replicas of blood vessels and kidneys, so-called organoids grown from human stem cells.
“We believe adding this enzyme copy, hrsACE2, lures the virus to attach itself to the copy instead of the actual cells,” Ali Mirazimi, adjunct professor at the Department of Laboratory Medicine at Karolinska Institutet and one of the study’s corresponding authors, says.
“It distracts the virus from infecting the cells to the same degree and should lead to a reduction in the growth of the virus in the lungs and other organs.”
The current study was limited to cell cultures and engineered miniature organs.
However, the same drug has already been tested against acute lung injury, acute respiratory distress syndrome and pulmonary arterial hypertension in clinical phase I and II studies.
The researchers note that the current study only examined the drug’s effect during the initial stages of infection and that further research is needed to determine if it is also effective during later stages of disease development.
ACE2 usually helps lungs and other organs to maintain normal function but when viruses bind to the protein they could damage the cells.
This could, according to the researchers, explain why some COVID-19 patients suffer severe lung disease and multi-organ failure.
In earlier work, Penninger and colleagues at the University of Toronto and the Institute of Molecular Biology in Vienna first identified ACE2, and found that in living organisms, ACE2 is the key receptor for SARS, the viral respiratory illness recognized as a global threat in 2003.
His laboratory also went on to link the protein to both cardiovascular disease and lung failure.
“Using organoids allows us to test in a very agile way treatments that are already being used for other diseases, or that are close to being validated.
In these moments in which time is short, human organoids save the time that we would spend to test a new drug in the human setting,” says Núria Montserrat, ICREA professor at the Institute for Bioengineering of Catalonia in Spain.
“The virus causing COVID-19 is a close sibling to the first SARS virus,” adds Penninger.
“Our previous work has helped to rapidly identify ACE2 as the entry gate for SARS-CoV-2, which explains a lot about the disease.
“This work stems from an amazing collaboration among academic researchers and companies, including STEMCELL Technologies in Vancouver, Nuria Montserrat in Spain, Drs. Haibo Zhang and Art Slutsky from Toronto and especially Ali Mirazimi’s infectious biology team in Sweden, who have been working tirelessly day and night for weeks to better understand the pathology of this disease and to provide breakthrough therapeutic options” says Penninger, professor in UBC’s faculty of medicine, director of the Life Sciences Institute and the Canada 150 Research Chair in Functional Genetics at UBC.
Several of the researchers report conflict of interests, including Josef Penninger who is a founder and shareholder in Apeiron Biologics. Please see the study for a complete list.
For more information
Cell
Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2
Vanessa Monteil, Hyesoo Kwon, Patricia Prado, Astrid Hagelkrüys, Reiner A. Wimmer, Martin Stahl, Alexandra Leopoldi, Elena Garreta, Carmen Hurtado del Pozo, Felipe Prosper, J.P. Romero, Gerald Wirnsberger, Haibo Zhang, Arthur S. Slutsky, Ryan Conder, Nuria Montserrat, Ali Mirazimi, Josef M. Penninger.
Link…
The University of British Columbia (UBC)
Link…
Karolinska Institutet
Link…
Karolinska University Hospital
Link…
National Veterinary Institute
Link…
IBEC
Pluripotency for Organ Regeneration, Institute for Bioengineering of Catalonia
Link…
BIST
Barcelona Institute of Science and Technology
Link…
IMBA
Institute of Molecular Biotechnology
Link…
STEMCELL Technologies Inc
Link…
CIMA
Center for Applied Medical Research University of Navarra,
Link…
Apeiron Biologics
Link…
STEMCELL Technologies Inc
Link…
Keenan Research Centre for Biomedical Science
Link…
ICREA
Catalan Institution for Research and Advanced Studies
Link…
Centro de Investigación Biomédica en Red en Bioingeniería, Biomateriales y Nanomedicina, 28029 Madrid, Spain
Link…
University of Toronto
Link…
Department of Medical Genetics, Life Science Institute, University of British Columbia, Vancouver
Link…
MDN
This post is also available in:
 Italian
Italian