Zucchero-Sugar
In mouse models of autoimmune disease, drinking sugary water worsened disease by activating inflammatory immune cells, a study led by Dr. Wanjun Chen from National Institute of Dental and Craniofacial Research, NIDCR discovered.
The findings suggest that a low-sugar diet may help prevent or treat autoimmune diseases such as Crohn’s disease and multiple sclerosis.
In recent decades, people in Western countries have been eating more food that is high in sugar, salt, and fat.
Experts think that diet may play a role in autoimmune diseases, which have also become more common in the West.
Autoimmune diseases arise when the immune system mistakenly attacks normal tissues in the body.
This response causes inflammation and gradual destruction of tissues.
For example, in Crohn’s disease, immune cells target the intestine.
In multiple sclerosis, the immune system eats away at the protective covering of nerves.
Scientists don’t have a clear idea of what causes autoimmunity, but some clues point to the role of diet.
High-salt intake promotes inflammation and worsens disease in animal models of multiple sclerosis.
To explore whether sugar affects autoimmunity, researchers led by Dr. Wanjun Chen from NIH’s National Institute of Dental and Craniofacial Research (NIDCR) studied mice with T cell (transfer colitis), a mouse model of Crohn’s disease and autoimmune encephalomyelitis, a model of multiple sclerosis.
 The researchers gave some of the mice regular water, while others received sugar-sweetened water at a dose similar to sugary drinks consumed by humans.
The researchers gave some of the mice regular water, while others received sugar-sweetened water at a dose similar to sugary drinks consumed by humans.
Compared to regular water, drinking sugary water worsened disease in both mouse models.
High sugar intake led to increased levels of an inflammatory immune cell called Th17 in and around the colons of mice with colitis and in the spinal cords and brains of mice with autoimmune encephalomyelitis.
Th17 cells are believed to play a role in tissue destruction in autoimmune diseases including Crohn’s disease and multiple sclerosis.
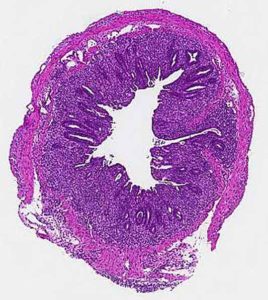
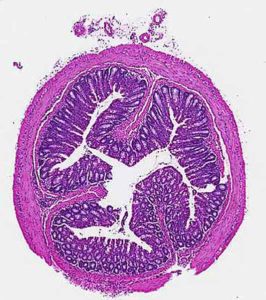
In mouse models of Crohn’s disease, drinking sugary water led to more severe destruction of colon tissue (dark purple in image on bottom), compared to mice that drank normal water (top).Wanjun Chen, NIDCR
To find out how sugar promotes Th17 cell activation, the scientists examined the behavior of immune cells grown in the lab.
Sugar appeared to trigger the activation of a molecule called transforming growth factor-ß (TGF-ß). TGF-ß, in turn, boosted levels of Th17 cells.
Blocking TGF-ß in the presence of sugar completely stopped production of Th17 cells.
The scientists observed a similar pattern in the mice with colitis.
Further experiments revealed that TGF-ß, which is normally in an inactive state, becomes activated in the presence of sugar via small chemicals called reactive oxygen species (ROS).
Environmental pollutants, tobacco smoke, radiation, certain drugs, and other agents can trigger production of ROS. Too much ROS can damage the body.
“We discovered a previously unrecognized mechanism by which high-sugar intake stimulates Th17 cell production and worsens autoimmunity in mice through ROS-driven activation of TGF-ß,” says Chen.
“The findings can inform research into whether a low-sugar diet could help prevent or treat certain autoimmune diseases, including Crohn’s disease and multiple sclerosis.”
For more information
Immunity
High glucose intake exacerbates autoimmunity through reactive oxygen species-mediated TGF-ß cytokine activation
Zhang D, Jin W, Wu R, Li J, Park S-A, Tu E, Zanvit P, Xu J, Liu O, Cain A, Chen W.
Link…
National Institute of Dental and Craniofacial Research (NIDCR)
Link…
The National Institutes of Health NIH
Link…
MDN
This post is also available in:
 Italian
Italian