|
Australian researchers have shed new light on the
nerve cell processes that lead to Alzheimer’s
disease (AD), overturning previously held ideas of
how the disease develops and opening the door to new
treatment options that could halt or slow its
progression. The study is published today in the
prestigious journal Science.
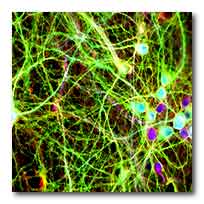
Cultured neurons. The colours highlight the human
tau protein in green, a structural component in red
and the DNA inside the cell nucleus in blue. Photo:
Supplied
Studying human brain tissue, the UNSW and
Neuroscience Research Australia research team
identified a protein, kinase p38γ, that is lost as
AD progresses. When they reintroduced the protein
into the brains of mice, it was shown to have a
protective effect against memory deficits associated
with the disease.
Two of the hallmarks of Alzheimer’s are the presence
of protein plaques (made up of amyloid-beta) and
tangles (made up of tau protein) in the brain. The
accumulation of these plaques and tangles is
associated with cell death, brain atrophy and memory
loss.
The research team has revealed that a crucial step
in the process that leads to tangles has been
misunderstood.
Previously, scientists believed the plaque-forming
protein, amyloid-beta, caused a modification –
called phosphorylation – to the tau protein
resulting in cell death and, ultimately, Alzheimer’s
disease. Increased phosphorylation of tau eventually
leads to its accumulation as tangles.

Results from the new study suggest that the
phosphorylation of tau initially has a protective
effect on neurons, and that amyloid-beta assaults
the protective functionality until it is
progressively lost.
This is the stage at which toxicity levels cause the
destruction of neurons and results in the cognitive
deficits associated with Alzheimer’s disease.
“Amyloid-beta induces toxicity in the neurons but
the first step in tau phosphorylation is actually to
decrease this toxicity,” said UNSW Professor Lars
Ittner.
“This is a completely new mindset; that the reason
tau becomes modified is actually to protect from
damage.”
The study used different mice models and human brain
tissue from the Sydney Brain Bank to identify a
protein called kinase p38γ, which assisted the
protective phosphorylation of tau and interfered
with the toxicity created by amyloid-beta.
“We used mice to screen for a very specific toxicity
that we knew from previous work is involved in the
progression of the disease,” said Professor Ittner.
“We set out to find mediators of this progression,
which led us quickly to our surprising finding. It
was the opposite of what we expected. It was only
when we changed our view of the process involved in
the development of AD that these results started to
make sense.”
Studying human brain tissue, Professor Ittner and
his team identified that p38γ is lost as AD
progresses, however a small amount does remain in
the brain.
“We found that p38γ, which initially offers
protection, fades away early in the brains of people
with AD, suggesting a loss of protection,” he said.
“Part of our study involved reintroducing p38? and
increasing its activity. We saw that, in mice, it
could prevent memory deficits from happening, so it
has true therapeutic potential. If we can stimulate
that activity, we may be able to delay or even halt
the progression of Alzheimer’s disease.”
The next step for the researchers will be to develop
their patented discoveries into a novel treatment
for humans, subject to new funding.
See also
Alzheimer: antibody reduces harmful brain amyloid
plaques (2016-09-02)
Link...
For more information
Science
Site-specific phosphorylation of tau inhibits
amyloid-ß toxicity in Alzheimer’s mice
Link...
UNSW Australia - The University of New South Wales
Link...
MDN |