|
A chemical that could potentially be used in eye
drops to reverse cataracts, the leading cause of
blindness, has been identified by a team of
scientists from UC San Francisco (UCSF), the
University of Michigan (U-M), and Washington
University in St. Louis (WUSTL).
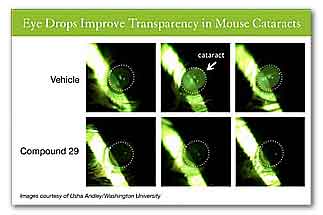
Mice, aged 5 weeks, were treated three times per
week for six weeks with "compound 29" in the right
eye and a vehicle control in the left eye. At the
end of the experiment, mice were examined by slit
lamp biomicroscopy, which showed improved
transparency. Dotted white lines highlight the edges
of the eye.
Identified as a “priority eye disease” by the World
Health Organization, cataracts — caused when the
lenses of the eyes lose their transparency — affect
more than 20 million people worldwide. Although
cataracts can be successfully removed with surgery,
this approach is expensive, and most individuals
blinded by severe cataracts in developing countries
go untreated.
Reported Nov. 5 in Science, the newly identified
compound is the first that is soluble enough to
potentially form the basis of a practical eye-drop
medication for cataracts.
Cataracts are primarily a disease of aging. As is
seen in neurodegenerative conditions such as
Alzheimer’s disease and Parkinson’s disease, a
hallmark of the condition is the misfolding and
clumping together of crucial proteins. In the case
of cataracts, the affected proteins are known as
crystallins.
Crystallins are the major component of fiber cells,
which form the eyes’ lenses, and the unique
properties of these cells make them particularly
susceptible to damage, said Jason Gestwicki, PhD,
associate professor of pharmaceutical chemistry at
UCSF and co-senior author of a paper on the new
research, most of which was undertaken in
Gestwicki’s laboratory at the U-M Life Sciences
Institute.
“Shortly after you’re born, all the fiber cells in
the eye lose the ability to make new proteins, or to
discard old proteins,” said Gestwicki, who has
continued his work on cataracts at UCSF, where he
joined the faculty about two years ago. “So the
crystallins you have in your eye as an adult are the
same as those you’re born with.”
In order for our lenses to function well, this
permanent, finite reservoir of crystallins must
maintain both the transparency of fiber cells and
their flexibility, as the eyes’ muscles constantly
stretch and relax the lens to allow us to focus on
objects at different distances.
The crystallins accomplish these duties with the
help of aptly named proteins known as chaperones,
which act “kind of like antifreeze,” Gestwicki said,
“keeping crystallins soluble in a delicate
equilibrium that’s in place for decades and
decades.”
This state-of-affairs is “delicate” because
pathological, clumped-together configurations of
crystallins are far more stable than properly
folded, healthy forms, and fiber-cell chaperones
must continually resist the strong tendency of
crystallins to clump.
A similar process underlies other disorders related
to aging, such as Alzheimer’s disease, but in each
of these diseases the specific protein that clumps
together and the place in the body that clumping
occurs is different. In all cases, these
clumped-together proteins are called amyloids.
In the new study, led by Leah N. Makley, PhD, and
Kathryn McMenimen, PhD, the scientific team
exploited a crucial difference between properly
folded crystallins and their amyloid forms: put
simply, amyloids are harder to melt.
The research group used a method known as
high-throughput differential scanning fluorimetry,
or HT-DSF, in which proteins emit light when they
reach their melting point. At the U-M Life Sciences
Institute’s Center for Chemical Genomics, the team
used HT-DSF to apply heat to amyloids while applying
thousands of chemical compounds.
Because the melting point of amyloids is higher than
that of normal crystallins, the team focused on
finding chemicals that lowered the melting point of
crystallin amyloids to the normal, healthy range.
The group began with 2,450 compounds, eventually
zeroing in on 12 that are members of a chemical
class known as sterols. One of these, known as
lanosterol, was shown to reverse cataracts in a July
paper in Nature, but because lanosterol has limited
solubility, the group who published that study had
to inject the compound into the eye for it to exert
its effects.
Using lanosterol and other sterols as a clue,
Gestwicki and his group assembled and tested 32
additional sterols, and eventually settled on one,
which they call “compound 29,” as the most likely
candidate that would be sufficiently soluble to be
used in cataract-dissolving eye drops.
In laboratory dish tests, the team confirmed that
compound 29 significantly stabilized crystallins and
prevented them from forming amyloids. They also
found that compound 29 dissolved amyloids that had
already formed. Through these experiments, said
Gestwicki, “we are starting to understand the
mechanism in detail. We know where compound 29
binds, and we are beginning to know exactly what
it’s doing.”
The team next tested compound 29 in an eye-drop
formulation in mice carrying mutations that make
them predisposed to cataracts. In experiments
conducted with Usha P. Andley, PhD, professor of
ophthalmology and visual sciences at WUSTL School of
Medicine, they found that the drops partially
restored transparency to mouse lenses affected by
cataracts, as measured by a slit-lamp test of the
sort used by ophthalmologists to measure cataracts
in humans.
Similar results were seen when compound 29 eye drops
were applied in mice that naturally developed
age-related cataracts, and also when the compound
was applied to human lens tissue affected by
cataracts that had been removed during surgery.
Gestwicki cautions that slit-lamp measures of lens
transparency used in the research are not a direct
measure of visual acuity, and that only clinical
trials in humans can establish the value of compound
29 as a cataract treatment. He has licensed the
compound from U-M, however, and Makley, a former
graduate student and postdoctoral fellow in the
Gestwicki laboratory, is founder and chief
scientific officer of ViewPoint Therapeutics, a
company that is actively developing compound 29 for
human use.
Dogs are also prone to developing cataracts. Half of
all dogs have cataracts by nine years of age, and
virtually all dogs develop them later in life. An
effective eye-drop medication could potentially
benefit about 70 million affected pet dogs in the
United States.
ViewPoint was formed through the “incubator” program
of the California Institute of Quantitative
Biosciences (QB3) at UCSF, and is located adjacent
to the UCSF campus in QB3’s Janssen Labs.
McMenimen, also a former graduate student in the
Gestwicki laboratory, is now the Claire Boothe Luce
Assistant Professor of Chemistry at Mt. Holyoke
College. The research was supported by the National
Institutes of Health, the American Foundation for
Pharmaceutical Education, and Research to Prevent
Blindness.
In addition to compound 29’s potential for cataract
treatment, the insights gained through the research
could have broader applications, said Gestwicki, a
member of UCSF’s Institute for Neurodegenerative
Diseases whose main research interest is dementia
and related disorders.
“If you look at an electron micrograph at the
protein aggregates that cause cataracts, you’d be
hard-pressed to tell them apart from those that
cause Alzheimer’s, Parkinson’s, or Huntington’s
diseases,” Gestwicki said. “By studying cataracts
we’ve been able to benchmark our technologies and to
show by proof-of-concept that these technologies
could also be used in nervous system diseases, to
lead us all the way from the first idea to a drug we
can test in clinical trials.”
For more information
Science
Pharmacological chaperone for a-crystallin partially
restores transparency in cataract models
Link...
Nature
Lanosterol reverses protein aggregation in cataracts
Link...
UC San Francisco (UCSF)
Link...
University of Michigan (U-M)
Link...
Washington University in St. Louis (WUSTL)
Link...
MDN |