|
New research from the University of North Carolina
School of Medicine for the first time explains
exactly how two brain regions interact to promote
emotionally motivated behaviors associated with
anxiety and reward.
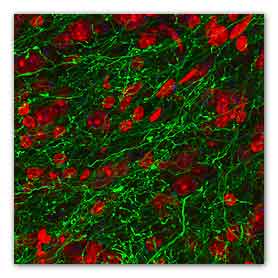
Microscopy image showing inhibitory fibers from the
BNST that innervate the VTA. Stimulation of these
fibers (green) produce rewarding and anti-anxiety
phenotypes in mice
The findings could lead to new mental health
therapies for disorders such as addiction, anxiety,
and depression. A report of the research was
published online by the journal, Nature, on March
20, 2013.
Located deep in the brain’s temporal lobe are
tightly packed clusters of brain cells in the
amygdala that are important for processing memory
and emotion.
When animals or people are in stressful situations,
neurons in an extended portion of the amygdala
called the bed nucleus of the stria terminalis, or
BNST, become hyperactive.
But, almost paradoxically, neurons in the BNST,
which modulate fear and anxiety, reach into a
portion of the midbrain that’s involved in
behavioral responses to reward, the ventral
tegmental area, or VTA.
“For many years it’s been known that dopamine
neurons in the VTA are involved in reward processing
and motivation.
For example, they’re activated during exposure to
drugs of abuse and naturally rewarding experiences,”
says study senior author Garret Stuber, PhD,
assistant professor in the departments of Psychiatry
and Cell Biology and Physiology, and the UNC
Neuroscience Center.
“On the one hand, you have this area of the brain –
the BNST – that’s associated with aversion and
anxiety, but it’s in direct communication with a
brain reward center. We wanted to figure out exactly
how these two brain regions interact to promote
different types of behavioral responses related to
anxiety and reward.”
In the past, researchers have tried to get a glimpse
into the inner workings of the brain using
electrical stimulation or drugs, but those
techniques couldn’t quickly and specifically change
only one type of cell or one type of connection. But
optogenetics, a technique that emerged about seven
years ago, can.
In the technique, scientists transfer
light-sensitive proteins called “opsins” – derived
from algae or bacteria that need light to grow –
into the mammalian brain cells they wish to study.
Then they shine laser beams onto the genetically
manipulated brain cells, either exciting or blocking
their activity with millisecond precision.
First, Stuber and colleagues used optogenetics for
“photo-tagging,” to optically identify different
types of neurons in vivo.
This enabled them to identify a neuron in the BNST
that’s projecting into the VTA. “So we know the
neuron is directly interfacing with a reward-related
brain region,” Stuber says.
They then exposed animals (mice) to a mild aversive
stimulus, a carefully controlled but
anxiety-provoking foot shock delivered repeatedly
and unpredictably.
The BNST neurons projecting into the VTA showed
changes in their firing rate, “But some cells would
increase their activity and other would suppress
their firing,” Stuber says, adding that it suggested
there are functionally distinct populations of
neurons within the BNST that are projecting to the
VTA, thus highlighting the complexity of this neural
circuit.
Stuber and his team then repeated the experiment,
but this time optically identified BNST neurons that
project to the VTA as either excitatory or
inhibitory cells, by integrating the approach they
developed with the use of transgenic animals that
allows for precise targeting of distinct neuronal
cell types.
The glutamate (excitatory) neurons were the cell
population that increased their activity in response
to the foot shocks.
And the GABAergic (inhibitory) cells showed activity
suppression during foot shock.
Finally, the researchers found that stimulating
either of these brain cell pathways had opposing
behavioral consequences.
The glutamate neurons provoked an aversive,
avoidance behavioral response and promoted
anxiety-like behavior in the mice.
In contrast, when Stuber’s team activated the
GABAergic pathway projections from the BNST into the
VTA, the animals showed reward-associated behaviors
and less anxiety. They preferred that stimulation
and would spend more time in the area of the cage
where they had received it.
“Because these cells are functionally and
genetically distinct from each other, our findings
also point to new potential targets for therapeutic
interventions in neuropsychiatric disorders
associated with alterations in motivated states such
as addiction.
Along with Stuber, UNC study co-authors from the
department of psychiatry are Joshua H. Jennings,
Dennis R. Sparta, Alice M. Stamatakis and Randall L.
Ung. Other co-authors on this study include Kristen
E. Pleil and Thomas L. Kash who are affiliated with
the department of pharmacology, and the Bowles
Center for Alcohol Studies.
Support for the study comes from the National
Institutes of Health, the Whitehall Foundation, and
the Foundation of Hope.
For more information:
nature
Distinct extended amygdala circuits for divergent
motivational states
Link...
Diverging neural pathways assemble a behavioural
state from separable features in anxiety
Link...
Anxiety is the sum of its parts
Link...
MDN |